Setting International Standards in
Analysing Patient-Reported Outcomes
and Quality of Life Endpoints
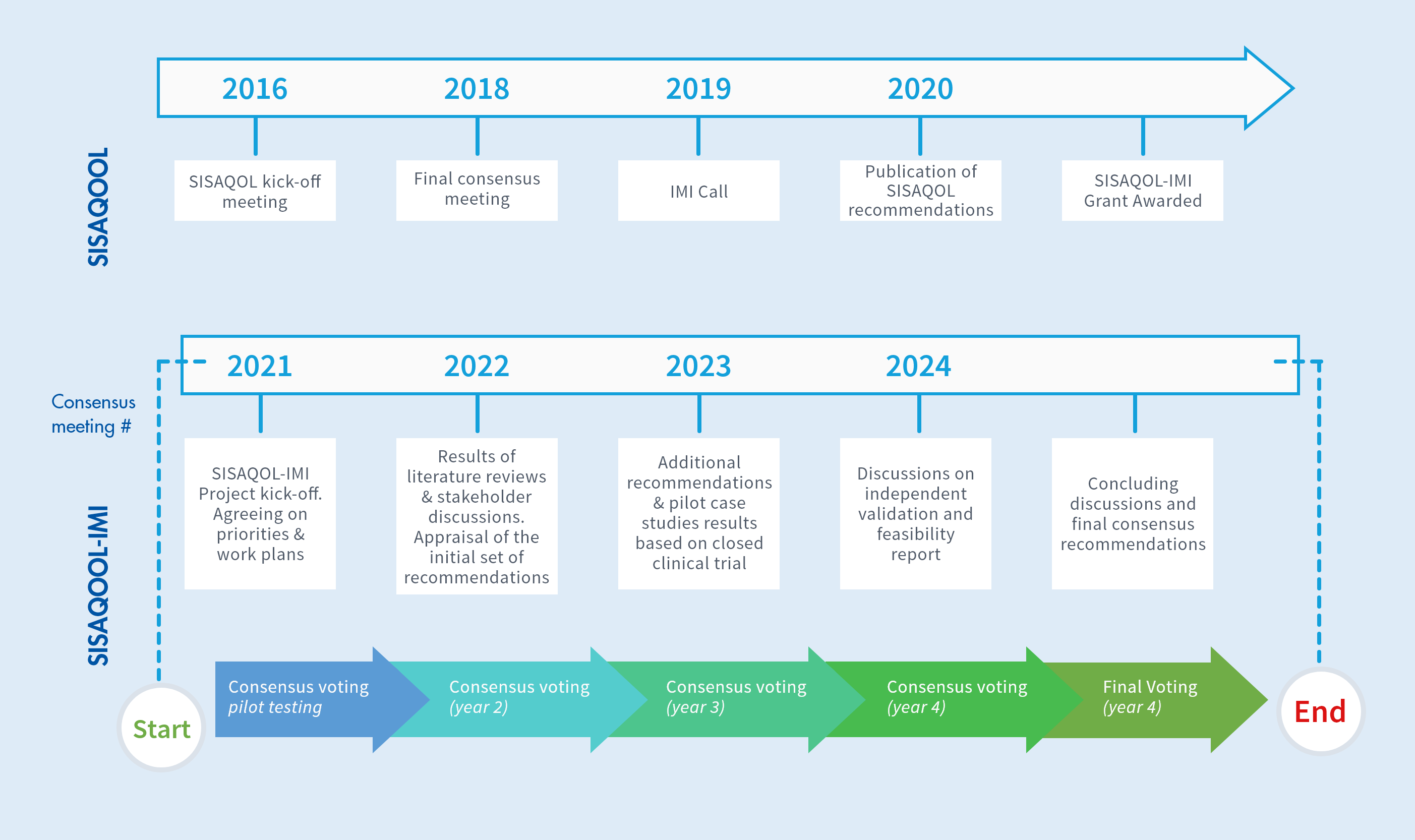
Project Timeline
SISAQOL-IMI launched in January 2021 and will run for four years and include 5 consensus meetings culminating in final consensus recommendations in 2025.
Key Work Package Milestones
| Milestone names | Related WPs | Due dates (in months) |
|---|---|---|
| Project kick-off meeting (Consensus meeting 1) | 1 | Due Date (in months): 2 |
| Consortium web-based workspace available to all partners | 1 | Due Date (in months): 1 |
| Members of independent scientific advisory board and ethics board involved | 1 | Due Date (in months): 3 |
| Consensus meetings organised (Consensus meetings 2 to 5) | 1 | Due Date (in months): 14, 26, 38, 44 |
| Stakeholder discussions and literature review report for RCTs | 2 | Due Date (in months): 14 |
| Initial RCT recommendations with pilot study results2 | 2 | Due Date (in months): 26 |
| Stakeholder discussions and literature | 3 | Due Date (in months): 14 |
| Initial Single-arm recommendations with pilot study results | 3 | Due Date (in months): 26 |
| Stakeholder discussion and literature review report for communication tools | 4 | Due Date (in months): 14 |
| Templates for graphical representation | 4 | Due Date (in months): 26 |
| Availability of dynamic webtool | 4 | Due Date (in months): 44 |
| Pilot and case studies identified | 5 | Due Date (in months): 6 |
| Case study database available on website | 5 | Due Date (in months): 46 |
| Stakeholder discussions and literature review report for clinically meaningful change | 6 | Due Date (in months): 14 |
| Template for consensus surveys | 7 | Due Date (in months): 6 |
| Proposed consensus statement drafted from relevant scientific WPs | 7 | Due Date (in months): 12, 22, 36, 42 |
| Consensus meeting reports | 7 | Due Date (in months): 3, 15, 27, 39, 45 |
| Final report on international consensus recommendations including a layperson version | 7 | Due Date (in months): 48 |
| Project website up and running | 8 | Due Date (in months): 6 |
| Educational workshop organised | 8 | Due Date (in months): 30 |
| Availability of educational tools and resources on | 8 | Due Date (in months): 44 |

